Latest Research
- 2017.02.01
- Nishiyama-Miura Group
Design of chemical linker and control of bioactivity for polymer-conjugated siRNA system
siRNA (small interfering RNA) is a double-stranded RNA composed of 20-22 base pairs. siRNA cleaves mRNA (messenger RNA) in the cell in a sequence-specific manner, leading to gene silencing with high selectivity [1]. Thus, siRNA is considered as a potent medicine due to the superior selectivity to the target, without a severe adverse effect.
Chemical modification of siRNA ameliorates its potency as a medicine; for example, facilitated internalization in the pathological lesion [2] and enhanced stability against enzymatic degradation in blood stream [3] have been addressed. Among them, conjugation with a functional polymer endows siRNA with multiple functionalities, because of easiness in functionalization of a polymer [4]. However, large volume of the conjugated polymer often inhibits siRNA recruitment into gene silencing pathway [5]. In general, siRNA forms RISC (RNA-induced silencing complex) with intracellular proteins, directed toward cleavage of mRNA with the complementary sequence; in this regard, the steric hindrance effect of the vicinal polymer hindered siRNA interaction with the RISC-related proteins, resulting in compromised gene silencing efficacy. Thus, an appropriate chemical linker that separates siRNA and the conjugated polymer selectively in the cell is often required.
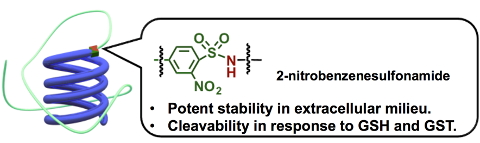 (Figure 1. Illustration of polymer-conjugated siRNA having 2-nitrobenzenesulfonamide group as a chemical linker)
(Figure 1. Illustration of polymer-conjugated siRNA having 2-nitrobenzenesulfonamide group as a chemical linker)
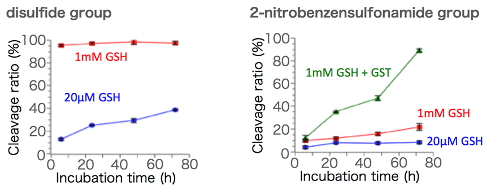 (Figure2. Cleavage ratio of the chemical linkers in response to reductive conditions)
(Figure2. Cleavage ratio of the chemical linkers in response to reductive conditions)
The higher redox potential in the cell relative to outside the cell is the most useful stimulus to design the chemical linker with intracellular cleavability. For example, the concentration of reductive glutathione (GSH) in the cell is ~1000 times higher relative to outside the cell. In this regard, disulfide group has been the principal chemical linker in response to the redox gradient across cellular membrane; however, disulfide group also suffers from moderate cleavage outside the cell, leading to increasing demand of another redox-sensitive chemical linker. To this end, we found that the utility of 2-nitrobenzenesulfonamide group as a chemical linker with responsiveness to redox potential (Figure 1). 2-nitrobenzenesulfonamide group originally works as a protecting group of primary/secondary amine, and can be cleaved by nucleophilic attack of thiol. In addition, glutathione-S-transferase (GST), which resides dominantly in the cell, enhances nucleophilicity of thiol group in GSH, potentially leading to selective cleavage of 2-nitrobenzenesulfonamide group in the cell. In fact, a polymer-conjugated siRNA having 2-nitrobenzenesulfonamide group exhibited higher stability in a mimicked extracellular environment (20 mM GSH), relative to disulfide system (Figure 2). Of note, moderate cleavage of 2-nitrobenzenesulfonamide group was observed in an mimicked intracellular GSH concentration (1 mM), which was accelerated by the co-existence of GST, suggesting the pivotal role of GST to cleave 2-nitrobenzenesulfonamide group. Considering the dominant presence of combination of GSH and GST, the cleavage of 2-nitrobenzenesulfonamide group is highly specific to intracellular condition. The intracellular cleavage of 2-nitrobenzenesulfonamide group was further confirmed by the effective induction of gene silencing for cultured cells, relative to polymer-conjugated siRNA having non-cleavable chemical linker. The utility of 2-nitrobenzenesulfonamide can be applied not only to a design of polymer-conjugated siRNA, but also to a design of various types of biomedical devices [6].
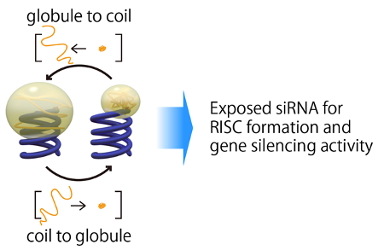 (Figure 3. Coil-globule transition of the conjugated polymer for control of siRNA bioactivity)
(Figure 3. Coil-globule transition of the conjugated polymer for control of siRNA bioactivity)
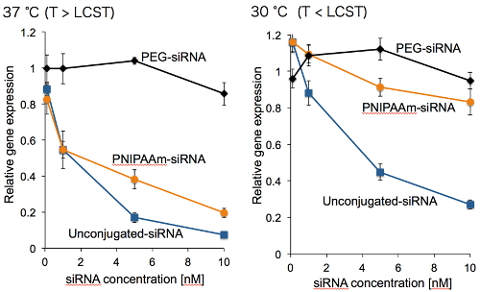 (Figure 4. Gene silencing efficacies of polymer-conjugated siRNA systems for cultured cells at two distinct temperatures)
(Figure 4. Gene silencing efficacies of polymer-conjugated siRNA systems for cultured cells at two distinct temperatures)
Meanwhile, steric hindrance effect of the conjugated polymer triggered our interest in developing a new methodology for a control of siRNA bioactivity in the cell; in situ regulation of the volume of the conjugated polymer allows controlled siRNA recruitment into gene silencing pathway, toward the localized therapy without undesired toxicity at normal tissues. In order to realize the artificial control of gene silencing activity, poly(N-isoproprylacrylamide) (PNIPAAm) was selected for conjugation with siRNA (Figure 3). PNIPAAm behaves as a coil form below LCST (lower critical solution temperature) of 33 °C, whereas shrinks into a globule form above LCST. This coil-globule transition associates with the considerable volume change and potentially leads to thermosensitive exposure of siRNA to Ago2 (Argonaute2), which plays a prime role in RISC formation. In fact, the synthesized PNIPAAm-siRNA exhibited compromised interaction with Ago2 below LCST (30 °C) and decreased gene silencing efficacy, whereas PNIPAAm-siRNA induced potent gene silencing efficacy above LCST (37 °C), which was close to the efficacy of unconjugated siRNA, based on the effective interaction with Ago2 (Figure 4). PEG-siRNA that does not show coil-globule transition in the aforementioned conditions did not induce gene silencing efficacy nor effective interaction with Ago2, confirming that coil-globule transition of the conjugated PNIPAAm is essential for the control of gene silencing activity of siRNA. As described, control of the steric hindrance of the vicinal polymer regulates the siRNA bioactivity, and this methodology would be translated into pathological lesions-specific induction of gene silencing effect, when the responsiveness to the targeted site-specific stimuli (e.g., pH and enzyme) is introduced into the conjugated polymer [7].
These studies suggest that a chemical design for circumvention of steric hindrance effect of the conjugated polymer is important for siRNA-conjugated polymer system; in addition, it is suggested that the steric hindrance effect can be utilized to control siRNA bioactivity based on coil-globule transition of the conjugated polymer. These systems are now in the study for in vivo experiments in order to examine the medicinal potential in a body.
References
[1] Elbashir, S. M.; Harborth, J.; Lendeckel, W.; Yalcin, a; Weber, K.; Tuschl, T. Nature 2001, 411, 494.
[2] Nair, J. K.; Willoughby, J. L.; Chan, A.; Charisse, K.; Alam, M. R.; Wang, Q.; Hoekstra, M.; Kandasamy, P.; Kel'in, A. V.; Milstein, S.; Taneja, N.; O'Shea, J.; Shaikh, S.; Zhang, L.; van der Sluis, R. J.; Jung, M. E.; Akinc, A.; Hutabarat, R.; Kuchimanchi, S.; Fitzgerald, K.; Zimmermann, T.; van Berkel, T. J.; Maier, M. A.; Rajeev, K. G.; Manoharan, M. J. Am. Chem. Soc. 2014, 136, 16958.
[3] Lee, S. H.; Mok, H.; Park, T. G. Macromol. Biosci. 2011, 11, 410.
[4] Rozema, D. B.; Lewis, D. L.; Wakefield, D. H.; Wong, S. C.; Klein, J. J.; Roesch, P. L.; Bertin, S. L.; Reppen, T. W.; Chu, Q.; Blokhin, A. V.; Hagstrom, J. E.; Wolff, J. A. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 12982.
[5] Takemoto, H.; Miyata, K.; Hattori, S.; Ishii, T.; Suma, T.; Uchida, S.; Nishiyama, N.; Kataoka, K. Angew. Chem., Int. Ed. 2013, 52, 6218.
[6] Huang, C. H.; Takemoto, H.; Nomoto, T.; Tomoda, K.; Matsui, M.; Nishiyama, N. ChemMedChem 2017, 12, 19.
[7] Harun, N. F. C.; Takemoto, H.; Nomoto, T.; Tomoda, K.; Matsui, M.; Nishiyama, N. Bioconjugate Chem. 2016, 27, 1961.



